Orlando, FL—Because of the way the vials are packaged, patients receiving filgrastim who weigh 60 kg to 85 kg often receive lower-than-recommended doses, without evidence of its noninferiority to recommended doses. Based on research presented at the 2018 National Comprehensive Cancer Network (NCCN) Annual Conference, this practice is not only acceptable, but cost-saving.
The recommended dose of filgrastim is 5 µg/kg. The commercially available vials of the drug come in 2 strengths— 300 µg and 480 µg. Because of these limitations in dosage formulations, clinicians frequently give a lower- than-recommended dose to patients weighing >60 kg in whom the ideal dose would be between 300 µg and 480 µg. In other words, some of the drug is wasted, explained Ahmed Elsayed, MD, MS, Hematology/Oncology, Joan C. Edwards School of Medicine, Edwards Comprehensive Cancer Center, Marshall University, Huntington, WV.
“It’s common practice in the oncology pharmacy community to use suboptimal dosing, but we do this without data to support it. Our analysis showed that for patients weighing up to 85 kg, it’s okay to give the ‘suboptimal’ dose….It’s ok to use one vial. But for patients over 85 kg, you should optimally dose,” Dr Elsayed said.
Lower-Dose Evaluation
Through chart review, Elsayed and colleagues identified 91 patients with chemotherapy-induced neutropenia who received 2 consecutive doses of filgrastim 300 µg. They divided the patients into 3 groups: low weight (<60 kg), medium weight (≥60 and <85 kg), and high weight (≥85 kg).
The researchers compared chemotherapy-induced neutropenia-related complications in patients with a higher weight (>60 kg), for whom 300 µg would be considered a suboptimal dose, versus patients weighing <60 kg, for whom a 300-µg dose would be recommended. The medium-weight and high-weight groups, who were presumably receiving subtherapeutic doses, were the cohorts of interest.
Outcomes and Cost-Savings
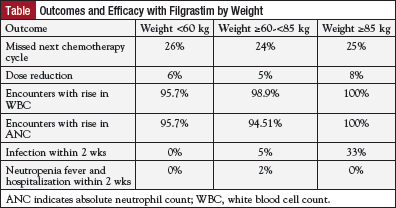
After administration of filgrastim, 98% of all patients had an increase in white blood cell count and 96% had an increased absolute neutrophil count. Similar responses for white blood cell and absolute neutrophil counts were observed among all 3 weight groups (Table).
The incidence of infections, delays in chemotherapy, and hospitalizations secondary to neutropenic fever were each approximately 5%. Meanwhile, chemotherapy dose reductions were necessary for 25% of patients. Patients in the medium-weight group did not have higher infection rates, but those in the high-weight group did have higher infection rates (5% vs 33%; P = .001).
“We showed that in patients [weighing] up to 85 kg, it’s okay to give the suboptimal dose, the one vial. Dosing filgrastim at 300 µg may be appropriate in this weight group,” Dr Elsayed concluded.
The investigators also concluded that this practice reduced the cost of treatment by 43%. Giving medium-weight patients 2 days of filgrastim at the lower dose (vs using the 480-µg vial) for 2 days translated into a savings of $147,274 in this one institution.