Acute myeloid leukemia (AML) is a rare but deadly hematologic cancer. In 2018, approximately 19,500 new cases of AML were diagnosed, and more than 10,600 people died from the disease in the United States.1 Although up to 70% of adults with AML have a complete response to initial treatment with cytotoxic chemotherapy, the responses are not durable.1 The 5-year survival rate for people with AML is only 24%.2
Since 2017, the management of AML in adults has undergone a transformation toward personalized medicine with the focus on patients with specific genetic mutations.3,4 In addition to cytogenetic testing, the measurement of minimal residual disease is now understood to have prognostic value in AML.3
After 40 years of no new drugs approved for the treatment of patients with AML, the US Food and Drug Administration (FDA) approved several novel targeted therapies, including midostaurin (Rydapt), an inhibitor of FMS-like tyrosine kinase 3 (FLT3); enasidenib (Idhifa), an IDH2 inhibitor; ivosidenib (Tibsovo), an IDH1 inhibitor; and gemtuzumab ozogamicin (Mylotarg), an antibody drug conjugate targeting CD33, for subtypes of patients with AML and a specific mutation.5-7
The identification of cytogenetic and molecular markers has improved our understanding of different prognostic cohorts in AML. Specifically, FLT3 gene mutations, which confer poor prognosis, are present in 25% to 30% of patients with AML.6 Midostaurin, the first FLT3 inhibitor approved by the FDA, has demonstrated safety and efficacy in combination with standard chemotherapy with cytarabine and daunorubicin induction and cytarabine consolidation in adults with newly diagnosed AML.6 It is hoped that second-generation FLT3 inhibitors will be more potent and selective.8
Xospata Approved as Monotherapy for AML with FLT3 Mutation
On November 28, 2018, the FDA approved oral gilteritinib (Xospata; Astellas Pharma), a tyrosine kinase inhibitor of several receptor kinases, including FLT3 mutation, for adults with relapsed or refractory AML and FLT3 mutation, as detected by an FDA-approved test.9,10 Gilteritinib is the first nonchemotherapy drug that can be used as monotherapy for this patient population.9
On the same day, the FDA approved an expanded indication for the LeukoStrat CDx FLT3 Mutation Assay, as a companion diagnostic to gilteritinib to detect FLT3 mutations in patients with AML before the administration of gilteritinib.9,10 The FDA reviewed gilteritinib using its fast track and priority review designations, and granted it an orphan drug status.9
“Approximately 25 to 30 percent of patients with AML have a mutation in the FLT3 gene,” said Richard Pazdur, MD, Director, FDA’s Oncology Center of Excellence. “These mutations are associated with a particularly aggressive form of the disease and a higher risk of relapse. Xospata targets this gene and is the first drug to be approved that can be used alone in treating patients with AML having a FLT3 mutation who have relapsed or who don’t respond to initial treatment,”9 he added.
Mechanism of Action
Gilteritinib is a small-molecule tyrosine kinase inhibitor that inhibits multiple receptor tyrosine kinases, including FLT3. In cells that exogenously express FLT3, including FLT3-ITD and tyrosine kinase domain mutations FLT3-D835Y and FLT3-ITD-D835Y, gilteritinib inhibits FLT3 receptor signaling and proliferation. Gilteritinib also induces cell death in leukemic cells that express FLT3-ITD.10
Dosing and Administration
Patients with relapsed AML should be selected for the consideration of gilteritinib treatment based on the presence of the FLT3 mutation in the blood or bone marrow, as detected by the FDA-approved LeukoStrat CDx FLT3 Mutation Assay.10
The recommended starting dose of gilteritinib is 120 mg (3 × 40 mg) orally once daily, with or without food. Because clinical response to gilteritinib can be delayed, treatment for a minimum of 6 months is recommended in patients with no disease progression or unacceptable toxicity.10
Gilteritinib tablets should not be broken or crushed, and they should be taken at approximately the same time each day.10
ADMIRAL Clinical Trial
The approval of gilteritinib is based on an interim analysis of the phase 3 ADMIRAL clinical trial, which included 138 adults (median age, 60 years) with relapsed or refractory AML who had a mutation in FLT3-ITD, D835, or I836, as identified or confirmed by the LeukoStrat CDx FLT3 Mutation Assay.9,10 Oral gilteritinib was administered at a dose of 120 mg once daily until disease progression or unacceptable toxicity. Dose changes were allowed to manage adverse events or to increase clinical benefit.9,10
Study participants had a median of 1 relapse (range, 0-2), 41% had disease that was refractory to previous therapy, and 20% had undergone stem-cell transplantation. The majority (77%) of patients were transfusion dependent, defined as requiring any platelets or red blood cells during the 56-day baseline period of the study.9,10
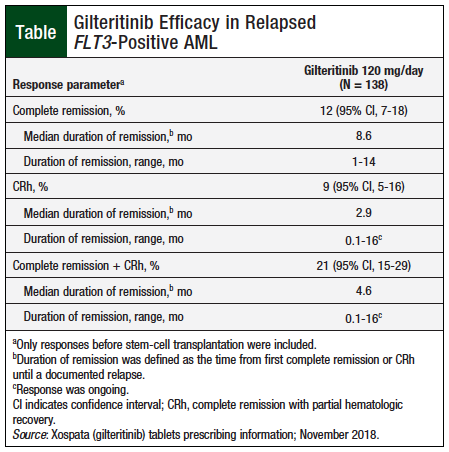
The key efficacy end points of the study were the rate of complete remissions and complete remission with partial hematologic recovery (CRh); secondary end points were duration of complete remission or CRh, and the rate of conversion from transfusion dependence to independence.10
The efficacy data for gilteritinib are summarized in the Table.10 Among the 106 patients who were dependent on red blood cell and/or platelet transfusions at baseline, 31% became transfusion independent during any of the 56-day period.10
Adverse Events
The safety profile of gilteritinib is based on data from 292 patients with relapsed or refractory AML who received a dose of 120 mg daily. The patients were exposed to gilteritinib for a median of 3 months (range, 0.1-43 months).10
The most common (≥20%) adverse reactions were myalgia or arthralgia (42%), an increase in transaminase (41%), fatigue or malaise (40%), fever (35%), noninfectious diarrhea (34%), dyspnea (34%), edema (34%), rash (30%), pneumonia (30%), nausea (27%), stomatitis (26%), and cough (25%).10
The most common (≥5%) serious adverse reactions were pneumonia (19%), sepsis (13%), and fever (13%).10
The permanent discontinuation of gilteritinib because of an adverse event was required in 8% of patients, with the most common events being pneumonia (2%), sepsis (2%), and dyspnea (1%).10
Contraindications
Gilteritinib is contraindicated in patients who have hypersensitivity to gilteritinib or any of its excipients. Anaphylactic reactions have occurred with gilteritinib in hypersensitive patients in clinical trials.
Drug Interactions
Gilteritinib should be avoided in patients who require combined P-gp and strong cytochrome (CY) P3A inducers.10
If the use of strong CYP3A inhibitors is considered essential, patients should be monitored for adverse reactions.10
The concomitant use of gilteritinib and drugs that target the 5-HT2B receptor or the sigma nonspecific receptor (eg, escitalopram, fluoxetine, sertraline) should be avoided.10
Use in Specific Populations
Gilteritinib can cause fetal harm. Females of reproductive potential should use effective contraception during treatment and for at least 6 months after the last dose of gilteritinib. Males of reproductive potential should use effective contraception during treatment and for at least 4 months after the last gilteritinib dose.10
Women should not breastfeed during treatment with gilteritinib and for at least 2 months after the last dose.10
The safety and effectiveness of gilteritinib have not been established in children. Among the 292 patients who received gilteritinib, 41% were aged ≥65 years. The efficacy and safety were similar in older and younger patients.10
Warnings and Precautions
Posterior reversible encephalopathy syndrome has been reported with gilteritinib. Symptoms, including seizure and altered mental status, resolve after gilteritinib discontinuation.10
The use of gilteritinib has been associated with prolonged cardiac ventricular repolarization. Electrocardiograms should be performed before the initiation of gilteritinib, on days 8 and 15 of cycle 1, and before the start of the next 2 cycles. The dosage of gilteritinib should be reduced in patients who have a corrected QT interval (by Fridericia formula) >500 msec. Hypokalemia or hypomagnesemia can increase the risk for QT prolongation and should be corrected before and during gilteritinib administration.10
Pancreatitis has been reported in clinical trials of gilteritinib. The dose of gilteritinib should be reduced or interrupted in patients with treatment-induced pancreatitis.10
Conclusion
With its approval, gilteritinib became the first and only FDA-approved targeted drug that can be used as monotherapy for patients with relapsed or refractory AML and the FLT3 mutation. Gilteritinib, an oral inhibitor of FLT3, has demonstrated complete remissions, durable remissions, and tolerability in patients with relapsed or refractory AML and an FLT3 mutation. In the phase 3 ADMIRAL clinical trial, gilteritinib demonstrated greater rates of complete remissions, including with CRh, longer duration of remission, and a high rate of conversion from transfusion dependence to transfusion independence. An ongoing phase 3 clinical trial is comparing the efficacy of gilteritinib and salvage chemotherapy in patients with relapsed or refractory AML and the FLT3 mutation.11
References
- National Cancer Institute. Adult acute myeloid leukemia treatment (PDQ)–health professional version. Updated February 7, 2018. www.cancer.gov/types/leukemia/hp/adult-aml-treatment-pdq. Accessed January 11, 2019.
- American Society of Clinical Oncology. Leukemia - acute myeloid - AML: statistics. www.cancer.net/cancer-types/leukemia-acute-myeloid-aml/statistics. Accessed January 11, 2019.
- Rowe JM. AML in 2017: advances in clinical practice. Best Pract Res Clin Haematol. 2017;30:283-286.
- Yang X, Wang J. Precision therapy for acute myeloid leukemia. J Hematol Oncol. 2018;11:3.
- National Cancer Institute. Two new therapies approved for acute myeloid leukemia. August 28, 2017. www.cancer.gov/news-events/cancer-currents-blog/2017/fda-enasidenib-vyxeos-aml. Accessed January 11, 2019.
- National Cancer Institute. Midostaurin approved by FDA for acute myeloid leukemia. June 1, 2017. www.cancer.gov/news-events/cancer-currents-blog/2017/fda-midostaurin-aml. Accessed January 11, 2019.
- US Food and Drug Administration. FDA approves Mylotarg for treatment of acute myeloid leukemia. September 1, 2017. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm574507.htm. Accessed January 11, 2019.
- Harris J. FLT3 inhibitors shake up treatment landscape for AML. Targeted Oncology. November 21, 2018. www.targetedonc.com/news/flt3-inhibitors-shake-up-treatment-landscape-for-aml. Accessed January 11, 2019.
- US Food and Drug Administration. FDA approves treatment for adult patients who have relapsed or refractory acute myeloid leukemia (AML) with a certain genetic mutation. November 28, 2018. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm627072.htm. Accessed January 11, 2019.
- Xospata (gilteritinib) tablets, for oral use [prescribing information]. Northbrook, IL: Astellas Pharma US; November 2018.
- ClinicalTrials.gov. A study of ASP2215 versus salvage chemotherapy in patients with relapsed or refractory acute myeloid leukemia (AML) with FMS-like tyrosine kinase (FLT3) mutation. https://clinicaltrials.gov/ct2/show/NCT02421939?cond=02421939&rank=1. Accessed January 11, 2019.