Biosimilars offer a significant opportunity for cost-savings yet present logistical hurdles for optimal implementation.1-4 Biosimilar savings nationwide are estimated to be $54 billion from 2017 to 2026, but these savings hinge on industry, regulatory, and policy changes to strengthen competition and sustainability in the biosimilar marketplace.5 The barriers to biosimilar adoption include provider acceptance, payer preferences, lack of an interchangeability designation for most biosimilars, and that biosimilars are assigned separate Healthcare Common Procedural Coding System codes requiring distinct build records within the electronic health record (EHR).1
A Biosimilar-First Pharmacist-Driven Strategy
At a multistate academic medical center, a biosimilar-first strategy starting in September 2020.1 The biosimilar-first strategy leveraged tools within the EHR to drive conversion from reference drugs to biosimilars—because guidance in the EHR is the standard approach when managing new drug coverage strategies—and to support pharmacist-driven therapeutic interchange.1,4 The objective of our approach was to transition the employee health plan population from reference drugs to biosimilars quickly and to realize cost-savings while minimizing the risk for therapy interruption, prior authorization burden, and the costs of conversion.
The preferred biosimilars were listed as the default drug or were listed first within ordering pathways and were noted as being preferred.1 The preferred biosimilars or reference drugs in this initiative included trastuzumab, rituximab, bevacizumab, filgrastim, epoetin alfa, and infliximab. Nonpreferred reference biologics and biosimilars were demoted in the drug list with a notation of being restricted to assist prescribers in appropriate drug selection.1 A pharmacist-driven therapeutic interchange authorized the pharmacy to change to a drug that is covered by the patient’s insurance if they did not cover the biosimilar that was ordered and to optimize biosimilars in the inpatient setting.1
Quarterly reports on utilization and health plan spending were generated from medical claims data for the reference and biosimilar drugs, absent of rebates. At baseline, a total of 434 patients received either the biosimilar or the reference drugs; biosimilar utilization by members was <3% across all drug groups, except for filgrastim, which had a 6.1% biosimilar utilization rate in the baseline time frame.
What We Achieved: Cost, Utilization, Relationships
Cost Savings
Over the course of the 2-year initiative, we achieved significant increases in biosimilar conversion rates and reduced drug spending by approximately $6 million for the employee health population. The savings were driven by rituximab ($2.5 million), trastuzumab ($1.2 million), bevacizumab ($1.1 million), infliximab ($1.1 million), and filgrastim ($100,000) market categories. For epoetin alfa, there was an increase in spending of $22,000 because of increased utilization.
Increased Utilization
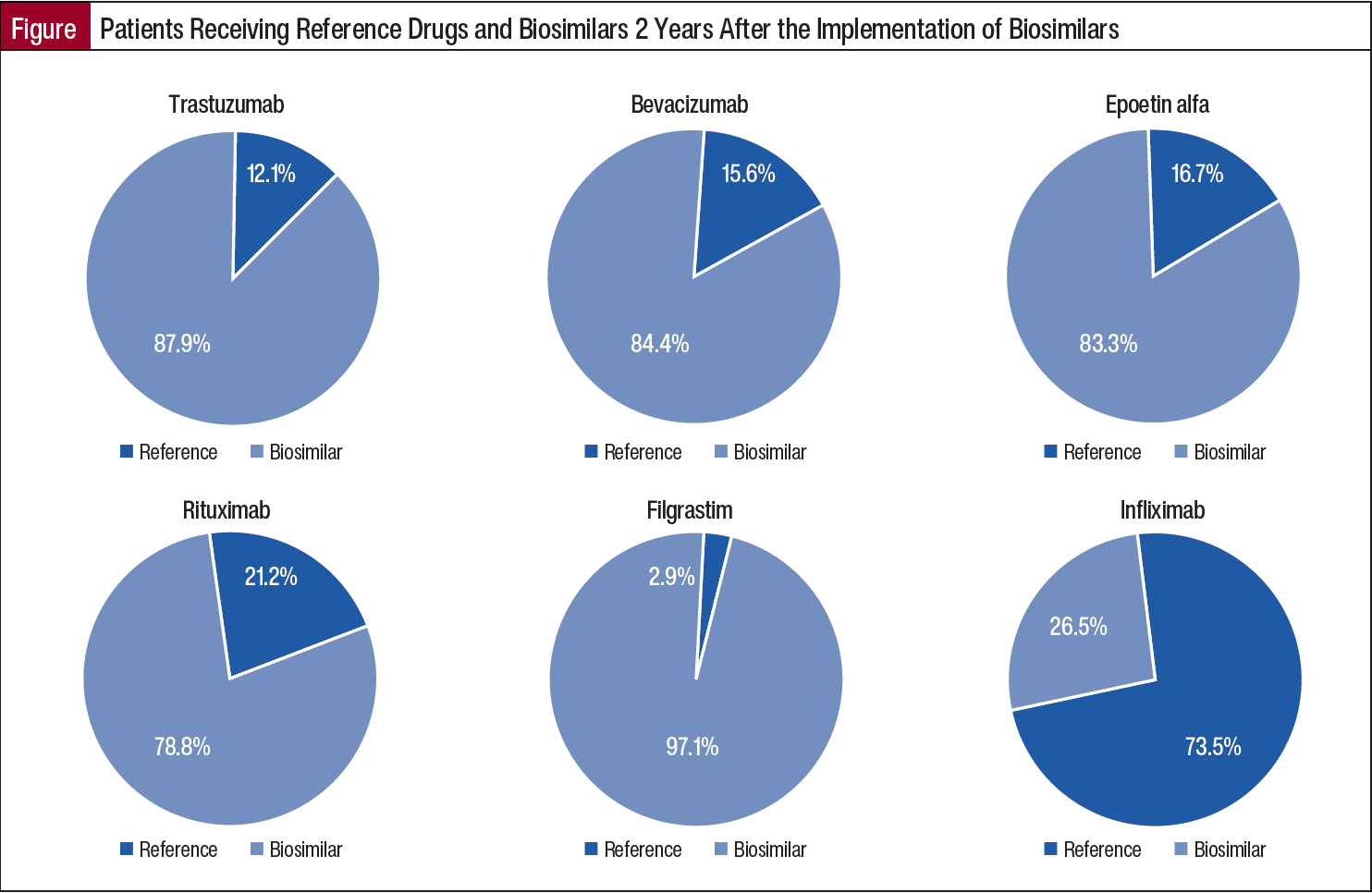
Similarly, after 2 years of implementation of biosimilars, the biosimilar utilization rate for members was 78.8% for rituximab; >80% each for trastuzumab, bevacizumab, and epoetin alfa drug groups; and 97.1% for filgrastim (Figure). For infliximab, where implementation followed a different timeline and process based on formulary adoption, 26.5% of members were receiving biosimilars at year 2.
Building and Strengthening Relationships
Building and strengthening relationships across a highly matrixed organization was crucial to the overall success of the utilization management strategy that was implemented. Educating multiple practice groups, explaining the primary goals of reducing drug expenditures, reducing inventory burden, and using reporting were key contributors to gaining buy-in throughout the conversion process.6
Closing Remarks
When compared with commercial industry benchmarks, our approach yielded improved or equivalent conversion to biosimilars across all market categories.7 We had greater success compared with benchmarks as a result of driving medication selection from reference drugs to biosimilars within the EHR, leveraging step-therapy prior authorization for reference drugs for bevacizumab and trastuzumab, having a multimodal approach for patients who remained on reference drugs 1 year after the implementation of biosimilars, and case-management approaches.7
Leveraging traditional and nontraditional utilization management techniques across a multidisciplinary team with shared goals and clear communication was an effective strategy to drive biosimilar adoption and reduce plan spending by 21% over 2 years at an academic medical center with a self-insured employee health plan population. For medical plan carriers or employee health plan populations, a combination of these utilization management techniques can be leveraged to promote biosimilar use and reduce healthcare costs.
Acknowledgments
The authors acknowledge Tina Seekamp, CPhT, for her assistance with the figures and graphs; Gary Effertz for the data extraction; and Ray Creighton-Lewis, PharmD, BCACP, BCPPS, AE-C, for coordination between the medical plan carrier and clinical teams and data analysis.
A longer version of this article with comprehensive data reporting was published in the Journal of Hematology Oncology Pharmacy, December 2024 Vol 14, No 6.
References
- Jensen CJ, Tichy EM, Lempke MB, et al. Implementing and optimizing biosimilar use at Mayo Clinic. Mayo Clin Proc. 2022;97:1086-1093.
- Webster J, Smith RE, Wieland D, et al. Cost savings of biosimilar pegfilgrastim in a Medicare OCM population. J Clin Oncol. 2020;38(15 suppl):e19362.
- Aschermann LM, Forshay CM, Kennerly-Shah J, Pilz J. The formulary process for biosimilar additions at a comprehensive cancer center. J Oncol Pharm Pract. 2022;28:185-189.
- Kar I, Kronz M, Kolychev E, et al. Biosimilar strategic implementation at a large health system. Am J Health Syst Pharm. 2022;79:268-275.
- Mulcahy AW, Hlavka JP, Case SR. Biosimilar cost savings in the United States: initial experience and future potential. Rand Health Q. 2018;7:3.
- Skerik S. The key to leading change: employee buy-in. Engagement Multiplier. Accessed September 9, 2024. www.engagementmultiplier.com/resources/the-key-to-leading-change-employee-buy-in/
- Magellan Rx Management. Medical Pharmacy Trend Report, Twelfth Edition. 2022. Accessed August 14, 2023. www1.magellanrx.com/documents/2022/12/medical-pharmacy-trend-report-2022.pdf