Disruptions to pharmaceutical supply chains have been ever-present, causing challenging shortages on key medicines.
Disruptions can result from a number of factors. For example, due to the complexity of the manufacturing processes for cancer drugs, which are antineoplastic and thus harmful to human cells, safety protocols are strict, and thus manufacturing and quality problems can occur. A related issue involves the balance between cost and the highly regulated manufacturing processes. Generic and older medicines are generally inexpensive, thus manufacturing margins are small, which further constrains manufacturing volume. A third issue is seen in the concentration of product manufacturing locations, as a number of medicines are each produced in a single facility.
Not surprisingly, manufacturing disruptions were exacerbated during the Covid-19 pandemic but are ongoing in many therapeutic areas. Currently, the American Society of Health-System Pharmacists (ASHP) has recently assessed active shortages at 277, which is down from its high of 323 in late 2023,1 yet new drug shortages continue to pose challenges for oncology care providers. New drug shortages across drug classes increased approximately 30% between 2021 and 2022.2 ASHP lists chemotherapy drugs among the top 5 active drug shortages by drug class as of September 30, 2024. (The remaining 4 classes include antimicrobial drugs, central nervous system agents, cardiology drugs, and hormone agents.)
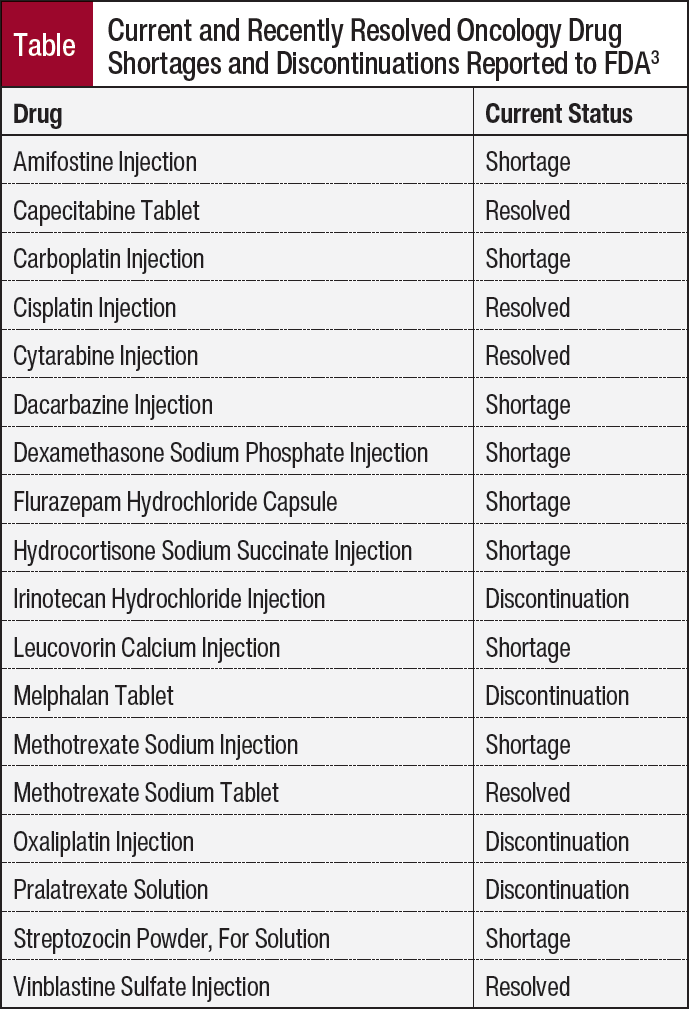
The FDA also monitors shortages and currently has 16 cancer drugs on their list of drug shortages (Table).3 The World Health Organization maintains a list of Essential Medicines that is updated every 2 years by the Expert Committee on Selection and Use of Essential Medicines, and 4 of the 20 essential cancer medicines are currently in shortage4:
- Cisplatin
- Carboplatin
- Capecitabine
- Dexamethasone
What Is Being Done?
The FDA works closely with drug manufacturers to prevent drug shortages or reduce their impact. Manufacturers are asked to notify the FDA of any coming shortages via the CDER Direct NextGen Portal (https://cdernextgenportal.fda.gov/Login_CDER) or email (
Ensuring Ongoing Patient Care
For active shortages, some hospitals may need to ration doses and use substitutions for standard of care in some patients. The American Society of Clinical Oncology, for example, has issued new clinical guidance on alternative treatments for patients with gastrointestinal cancers commonly treated with carboplatin and cisplatin.5 In addition, ASHP encourages healthcare providers and their distributors to maintain a buffer supply of critical medicines to mitigate the impact of supply chain disruptions on patient care.6
Changes in National Policy
On a national policy level, the US Congress is attempting to address supply chain issues resulting from a concentration of manufacturing facilities. In the generic medicines sphere, for example, 47% of the US supply is imported from India.7 In July 2021, the Biden administration issued its National Strategy for a Resilient Public Health Supply Chain, which is aimed at building a more robust domestic manufacturing base.7 In addition to India, China also has a large source of consolidated manufacturing facilities. The BIOSECURE Act, which is poised for Senate action after receiving approval in the House, prohibits firms that receive funds from the US government from working with 5 Chinese “companies of concern.” This act takes effect as of January 1, 2032, which is intended to allow US manufacturers ample time to unwind operations with these 5 firms.7
Drug shortages have implications for patients as well as the healthcare system. For healthcare institutions, increased costs can be a substantial burden, but these increased costs are not limited to the institutions alone—patients also experience increased out-of-pocket costs. In addition, patients may be exposed to higher risk for adverse events or suboptimal response as a result of substitutions for standard-of-care treatments.8
Adjustments in clinical care and the establishment of policy imperatives are expected to mitigate drug shortages in oncology and elsewhere, and TOP will monitor ongoing actions from national as well as local perspectives.
References
- American Society of Health-System Pharmacists. National drug shortages January 2001 - September 2024. Accessed December 17, 2024. www.ashp.org/-/media/assets/drug-shortages/docs/2024/2024-Drug-Shortages-Survey.pdf
- USP. Why cancer medicines are and continue to be vulnerable to drug shortages. Accessed December 17, 2024. https://qualitymatters.usp.org/why-cancer-medicines-are-and-continue-be-vulnerable-drug-shortages
- Food and Drug Administration. Drug Shortages. Accessed December 17, 2024. https://www.fda.gov/drugs/drug-safety-and-availability/drug-shortages
- World Health Organization. WHO Model Lists of Essential Medicines. Accessed December 17, 2024. https://www.who.int/groups/expert-committee-on-selection-and-use-of-essential-medicines/essential-medicines-lists
- American Society of Clinical Oncology. Gastrointestinal Cancer Guidance. Accessed January 3, 2025. https://society.asco.org/sites/new-www.asco.org/files/content-files/advocacy-and-policy/documents/2023-GI-ds-recs.pdf
- American Society of Health-System Pharmacists. Policy Solutions to Address the Drug Shortage Crisis. Accessed January 3, 2025. https://www.ashp.org/-/media/assets/advocacy-issues/docs/2023/ASHP-Drug-Shortage-Recommendations.pdf
- Center for Strategic & International Studies. A Bilateral Approach to Address Vulnerability in the Pharmaceutical Supply Chain. Accessed January 3, 2025. https://www.csis.org/analysis/bilateral-approach-address-vulnerability-pharmaceutical-supply-chain
- Phuong JM, Penm J, Chaar B, et al. The impacts of medication shortages on patient outcomes: a scoping review. PLoS One. 2019;14(5):e0215837.